| IRB NUMBER | INVESTIGATORS | STUDY CONTACT |
| HSC-SPH-19-0406 | Sarah E. Messiah, PhD, MPH, FTOS
UTHealth Houston | Sarah E. Barlow, MD
Children's Health
UT Southwestern | Faisal Qureshi, MD
Children's Health
UT Southwestern | Rashon King, MPH
Rashon.M.Braxton@uth.tmc.edu |
Invitation to take part in a research study
Dr. Sarah E. Messiah is inviting you to take part in a research study. You need to know about the study so you can decide if you would like to join the study or not. If you enter the study, you will be asked to sign this form. You may take this paper home to talk with your family and friends before making your decision.
Your parent(s) or legal guardian will also be asked to give their permission for you to take part in this study. Please talk this over with your parent(s) or legal guardian before you decide whether or not to be in the study.
Who is eligible for this study?
To be eligible, you must:
- Be between 12-18 years of age.
- Have parent/guardian permission to participate if you are a minor.
- Be a patient referred for bariatric surgery at Children's Health.
- Not have diabetes that requires taking mealtime insulin.
- Have a Facebook account to access the healthy lifestyle intervention.
Why is this study being done?
We are inviting you to take part in a research study because we are trying to learn more about your decision to have weight loss surgery, and what online tools such as social media, blogs, online chatrooms and apps, etc. that you would be interested in using to help with your weight loss journey before and after surgery.
Why am I being asked to take part in this research study?
We are inviting you to take part in this research study because you have decided to have bariatric surgery.
How many people will take part in this study?
About 20 adolescents ages 12-18 will be in this study. This study will be done at about four Children's Health sites in the Dallas-Fort Worth area.
What is involved in the study?
If you agree to be in this study, the following things will happen:
- You must consent to participate in the study.
- You will be in the study for 18 months.
- You will have up to four follow-ups in total. One before surgery, and the remaining follow-ups after the surgery.
- You will be asked to spend a total of up to 26 hours in the study. Before surgery, you will be asked to participate in online educational sessions to learn more about weight loss surgery. After you have your surgery, we will ask you to participate in online educational sessions where you will be provided with information and tools to help you on your weight loss journey.
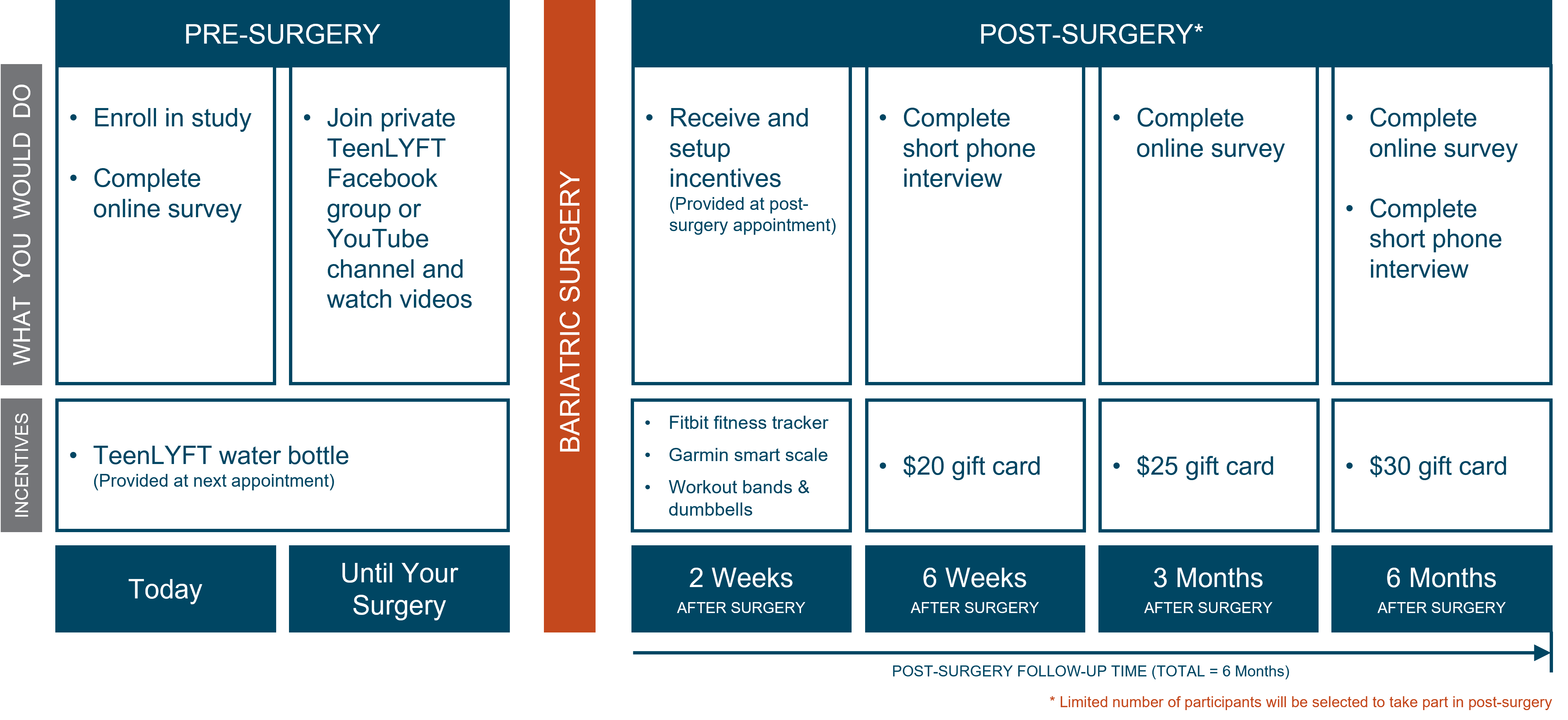
How long will this take?
You will be in the study for 18 months. During this time you will be asked to participate in follow-up surveys and take part in educational content that will support your weight loss journey. The total amount of time you will spend in the study is 26 hours.
What are the benefits of taking part in this study?
Taking part in this study may or may not provide you with support for weight loss. Information from this study might help researchers to come up with programs that can provide support before and after weight loss surgery.
What are some of the risks and discomforts?
- Psychological Stress: The risk of taking part in this study are minimal. At times you may feel awkward or embarrassed while answering the questions. However, you do not have to answer questions that you are not comfortable answering.
- Loss of Confidentiality: Any time information is collected; there is a potential risk of loss of confidentiality. Every effort will be made to keep your information confidential; however, this cannot be guaranteed.
- Other Risks: If you suffer an injury as a result of taking part in this research study, please understand that nothing has been arranged to provide free treatment of the injury or any other type of payment. However, necessary facilities, emergency treatment, and professional services will be available to you, just as they are to the general community. You should report any such injury to the Principal Investigator Dr. Sarah E. Barlow at 214-456-5466. You will not give up any of your legal rights by signing this consent form.
What options are available if I decide not to take part in this research study?
You will continue in the bariatric surgery program without study participation.
Can I stop taking part in this study?
You may decide to stop taking part in the study at any time. To withdraw from the study, please contact the Principal Investigator Dr. Sarah E. Barlow at 214-456-5466.
Your doctor or the sponsor can stop the study at any time. Your doctor or the sponsor may stop your participation in the study if you do not meet all the requirements of the study, or if the study is not in your best interest.
If you stop participating in this study, the information already collected will still be used in the data analysis. However, no further information will be collected without your permission.
While taking part in this study, the study team will notify you of new information that may become available and could affect your willingness to stay in the study.
Is there a cost to be in the study?
There is no cost to take part in the study.
Will I be paid if I take part in this research study?
You may receive a total of 3 gift cards over the 18-month period that you are enrolled in the study. The gift cards may be provided at 3 specific clinical visits. The first gift card of $20 may be provided to you at your 6-week post-operative clinical visit, the second gift card of $25 at the 3-month post-operative clinical visit, and lastly, the third gift card of $30 at the 6-month post-operative clinical visit.
You may also receive a Fitbit fitness tracker, a smart scale, resistance bands, dumbbell set, and a water bottle. You may be provided a water bottle at your first visit. Upon completion of your surgery you may recieve the fitness tracker, the smart scale, resistance bands, and dumbbells to aid in your weight loss journey while in the study. Please note that the technology provided to you are not medical devices.
Will my information be kept confidential?
Your privacy is important and your participation in this study will be kept confidential. However, absolute confidentiality cannot be guaranteed.
If you sign this document, you give permission to UTHealth Houston, UT Southwestern, and Children's Health to use and disclose (release) your health information. The health information that we may use or disclose for this research includes medical records, medical history, nutrition, exercise behavior, lab tests pertaining to bariatric surgery, and obesity-related comorbidities. Please understand that health information used and disclosed may include information relating to behavioral health, and psychiatric care.
Personal identifiers such as your name and medical record number will be removed from the information and samples collected in this study. After we remove all identifiers, the information or samples may be used for future research or shared with other researchers without your additional permission.
Representatives of the organizations listed below will see your and other personal identifiers when they review your research records and medical records for the purposes of verifying study data:
- Representatives of UTHealth Houston
- Representatives of UT Southwestern
- Representatives of Children's Health
What if you have any questions?
You can ask questions at any time. You can ask now, or you can ask later. You can talk to the study doctor or you can talk to the study coordinator Rashon King. If you would like to talk to the study coordinator, she can be contacted at 972-546-2958.
The Committee for Protection of Human Subjects at the University of Texas Health Science Center has reviewed this research study. You may contact them for any questions about your rights as a research subject, and to discuss any concerns, comments, or complaints about taking part in a research study at 713-500-7943.